ATSC 113 Weather for Sailing, Flying & Snow Sports
Standard Atmosphere - Pressure and Density
Learning Goal 2a. Draw and describe the variation of pressure & density with altitude
If you have ever climbed a mountain, you know that the air gets thinner, thus making it harder to breathe (lower density at higher altitudes). If you've been in a fast elevator in a tall building, your ears might pop due to the pressure change (lower pressure at higher altitudes).
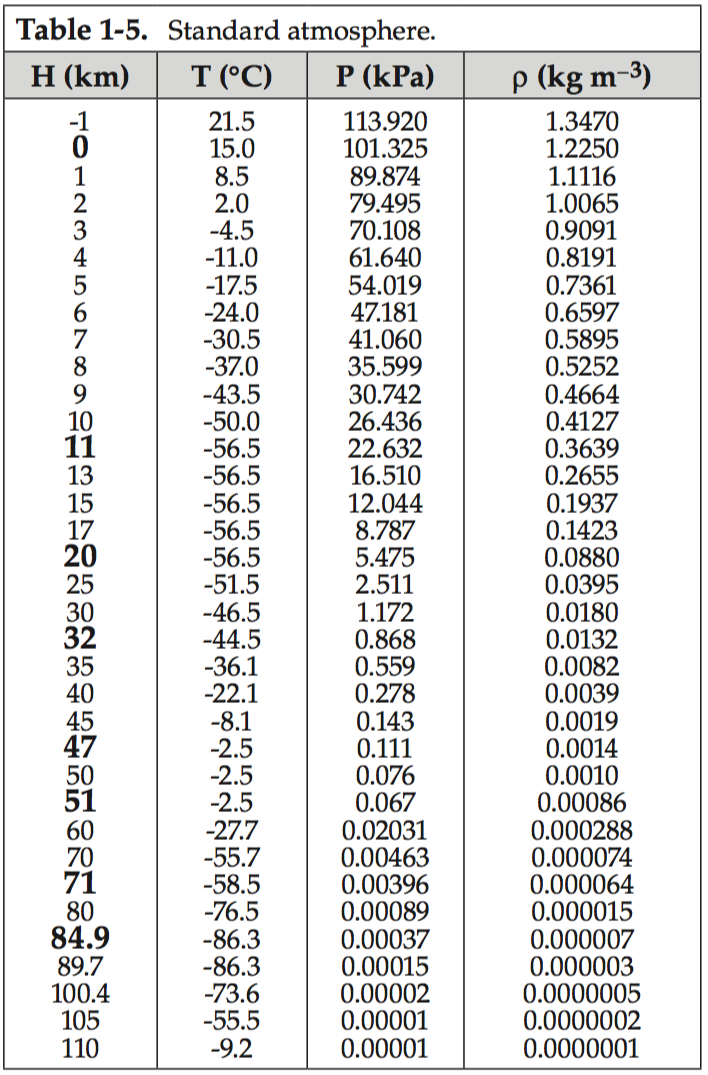
This average variation of air pressure (P), temperature (T), and density (ρ) with altitude is called the standard atmosphere. The table below shows how those variables change with height (H) above sea level. Temperature is discussed in more detail in Learning Goal 3a.
Pressure and Density
Pressure is measured with a barometer. It indicates the amount of force that the air is pushing against each square meter of an object (such as your body, or the mechanism in an aircraft altimeter). Greater pressure implies greater force per unit area. It has units of Pascals (Pa). Normal atmospheric pressure at sea level is about 101,000 Pa; namely 101 kPa (kiloPascals). You can see the effect of atmospheric pressure when you try to suck the air out of a thin-walled plastic bottle - - the atmospheric pressure outside the bottle pushes the sides of the bottle inward, crushing the bottle.
It is not so easy to measure air density - - most households and
most aircraft don't have sensors that measure this (instead, it is
calculated from pressure and temperature). Density is the amount
of mass of air in each cubic meter of volume. It has units of
kilograms of air per cubic meter. Think of it as the mass of air
inside an empty cardboard box of dimensions 1 x 1 x 1 m. Typical
air density at sea level is about 1.225 kg / m3 . But
air density strongly affects aircraft flight - - at higher altitudes
where the air is thin, aircraft engines make less power and propellors
are less effective and wings create less lift. These issues are
discussed in Learning Goal 2c on "density altitude".
The table above shows that pressure decreases smoothly with altitude (H), as does air density. Note, both z and H mean the same thing here; namely, height above mean sea level. Pilots might refer to this as altitude. At sea level (z = 0), average pressure is about 101 kiloPascals (kPa), and average air density is about 1.225 kg/m3. As you go to higher altitudes above sea level, the pressure and air density decrease rapidly with increasing height at first, and then decrease more slowly with height at higher altitudes.
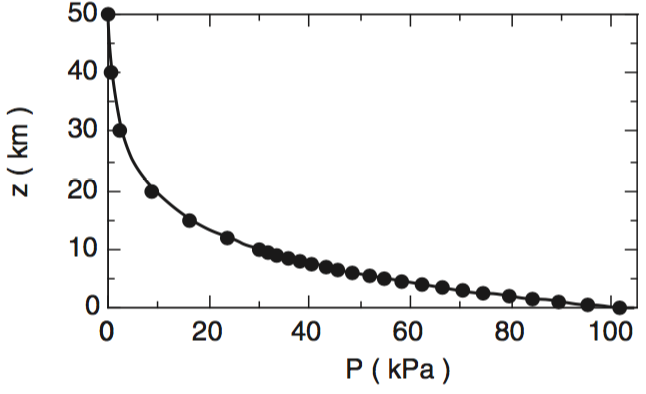
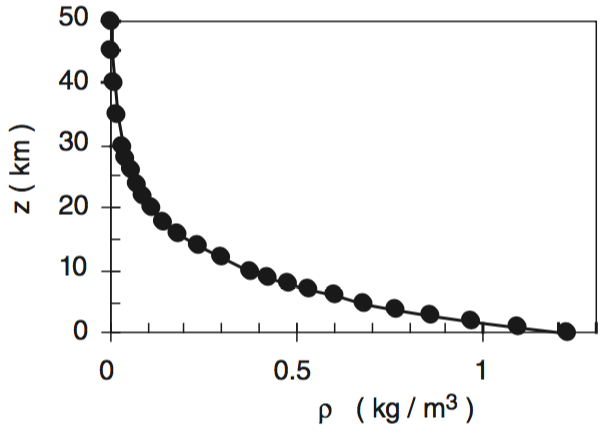
This smooth variation can also be seen in graphs of pressure (P) vs. altitude (z), and plots of density (ρ) vs. altitude (z), as plotted below. By the time you get to an altitude of 40 km, the air is so thin that pressure and density are near zero.
 |
 |
Nonetheless, the depth of the atmosphere is thin compared to the
diameter of the earth as observed by NASA astronauts. In the
first photo below, taken at sunrise, the atmosphere is the thin white
line around the surface of the earth. In the second photo, a
different sunrise and a zoomed view, the 40 km depth is roughly the
thickness of the red/yellow layer of air touching the earth (the earth
is in silhouette; it is the black area in the bottom quarter of the
photo).
— Images from NASA
Note: Info about temperatures at different altitudes in a standard atmosphere are discussed in Learning Goal 3a.
Key word: standard atmosphere
Extra info for experts; not needed for this course.
The International Standard Atmosphere (ISA): Wikipedia.
The US Standard Atmosphere: Wikipedia.
The US Naval Research Lab atmosphere model from the ground to space, for estimating air drag on satellites Wikipedia.
Image credits. All the drawings were made by Roland Stull, and all are copyright by him and used with his permission.